
The curve between the critical point and the triple point shows the ethanol boiling point with changes in pressure. The phase diagram for ethanol shows the phase behavior with changes in temperature and pressure. 
However, at low temperature and/or very high pressures it becomes a solid. See also more about atmospheric pressure, and STP - Standard Temperature and Pressure & NTP - Normal Temperature and Pressure,Īs well as Thermophysical properties of: Acetone, Acetylene, Air, Ammonia, Argon, Benzene, Butane, Carbon dioxide, Carbon monoxide, Ethane, Ethylene, Helium, Hydrogen, Hydrogen sulfide, Methane, Methanol, Nitrogen, Oxygen, Pentane, Propane, Toluene, Water and Heavy water, D 2O.Įthanol is a liquid at standard conditions. Specific Heat (Heat Capacity), C p and C v.Specific Gravity (liquid) (relativ to water)įollow the links below to get values for the listed properties of ethanol at varying pressure and temperature: Log K OW (Octanol/Water Partition Coefficient) Specific heat capacity, Cv (isochoric) (gas) Specific heat capacity, Cp (isobaric) (gas) Values are given for liquid at 25 oC /77 oF / 298 K and 1 bara, if not other phase, temperature or pressure given.įor full table with Imperial Units - rotate the screen! Property The phase diagram of ethanol is shown below the table.Ĭhemical, physical and thermal properties of ethanol : Ethanol is also used as a clean-burning fuel source. The compound is widely used as a chemical solvent, either for scientific chemical testing or in synthesis of other organic compounds. 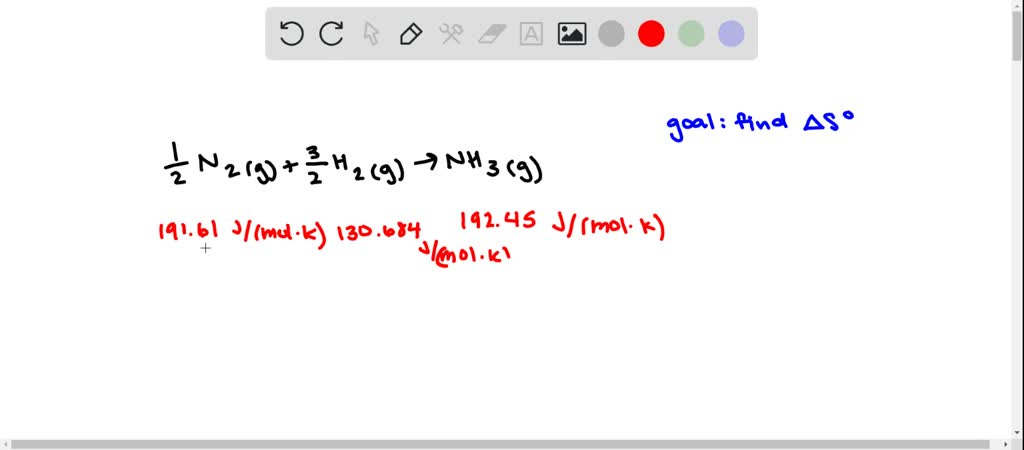
It also has medical applications as an antiseptic and disinfectant. It is a psychoactive substance and is the principal type of alcohol found in alcoholic drinks. It is produced via petrochemical processes or naturally by the fermentation of sugars by yeasts.Įthanol is most commonly consumed as a popular recreational drug. The standard entropy of reaction helps determine whether the reaction will take place spontaneously.Ethanol (Ethyl Alcohol), C 2H 5OH, is a volatile, flammable, colorless liquid with a slight characteristic odor. ΔS° rxn = S o(products) - S o (reactants) Chemical equations make use of the standard molar entropy of reactants and products to find the standard entropy of reaction: The heat capacity of the gas from the boiling point to room temperature.Ĭhanges in entropy are associated with phase transitions and chemical reactions.The latent heat of vaporization of the liquid.The heat capacity of the liquid from the melting point to the boiling point.The latent heat of fusion of the solid.The heat capacity of one mole of the solid from 0 K to the melting point (including heat absorbed in any changes between different crystal structures).The standard molar entropy of a gas at STP includes contributions from: The total molar entropy is the sum of many small changes in molar entropy, where each small change can be considered a reversible process. Here, dq k/T represents a very small exchange of heat energy at temperature T. If a mole of substance were at 0 K, then warmed by its surroundings to 298 K, its total molar entropy would be the addition of all N individual contributions:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
